Archives
- 2026-05
- 2026-04
- 2026-03
- 2026-02
- 2026-01
- 2025-12
- 2025-11
- 2025-10
- 2025-09
- 2025-03
- 2025-02
- 2025-01
- 2024-12
- 2024-11
- 2024-10
- 2024-09
- 2024-08
- 2024-07
- 2024-06
- 2024-05
- 2024-04
- 2024-03
- 2024-02
- 2024-01
- 2023-12
- 2023-11
- 2023-10
- 2023-09
- 2023-08
- 2023-07
- 2023-06
- 2023-05
- 2023-04
- 2023-03
- 2023-02
- 2023-01
- 2022-12
- 2022-11
- 2022-10
- 2022-09
- 2022-08
- 2022-07
- 2022-06
- 2022-05
- 2022-04
- 2022-03
- 2022-02
- 2022-01
- 2021-12
- 2021-11
- 2021-10
- 2021-09
- 2021-08
- 2021-07
- 2021-06
- 2021-05
- 2021-04
- 2021-03
- 2021-02
- 2021-01
- 2020-12
- 2020-11
- 2020-10
- 2020-09
- 2020-08
- 2020-07
- 2020-06
- 2020-05
- 2020-04
- 2020-03
- 2020-02
- 2020-01
- 2019-12
- 2019-11
- 2019-10
- 2019-09
- 2019-08
- 2019-07
- 2019-06
- 2019-05
- 2019-04
- 2018-11
- 2018-10
- 2018-07
-
The focus of this review is on the
2020-01-06

The focus of this review is on the diacylglycerol acyltransferase (DGAT, EC 2.3.1.20) enzymes, DGAT1 and DGAT2, that catalyze the esterification of a fatty acyl moiety to a DAG molecule to produce TAG. These enzymes commit DAG to being stored as TAG rather than being diverted for the synthesis of ph
-
Since protein kinase CK has over known substrates we
2020-01-06
Since protein kinase CK2 has over 300 known substrates we examined if the inhibitory effects described above could also be detected using another substrate. Therefore, we employed the synthetic peptide RRRADDSDDDDD which is often used in CK2 inhibitory studies. Quite contrary to the phosphorylation
-
In rodent models the loss of estrogens through ovariectomy h
2020-01-06

In rodent models, the loss of estrogens through ovariectomy has been shown to promote the development of intracranial aneurysms. Conversely, the administration of an ruthenium red receptor beta agonist restores its protective effect, but is abolished with the introduction of a nitric oxide synthase
-
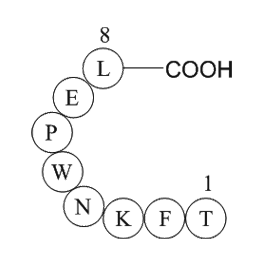
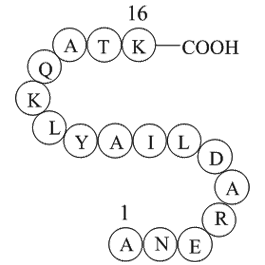
GSK 5959 receptor Compounds were synthesized using a
2020-01-06
Compounds were synthesized using a facile 8-step convergent synthesis ( and ). Briefly, (Boc)cyclen (compound ) was coupled using TBTU to Fmoc- or Cbz-protected amino alkyl acids (–), followed by deprotection using 20% piperidine in DMF, or H on Pd/C, respectively (–) (). Deprotected linkers were co
-
Introduction Prolyl hydroxylation is a widely observed
2020-01-06
Introduction Prolyl hydroxylation is a widely observed post translational modification in collagen, an abundant animal protein. The high content (~13% per chain of the triple helical structure) of 4-hydroxyproline (Hyp,O) in collagen, togather with the natural abundance of collagen has led to the e
-
Phosphorylation of small GTPases has been
2020-01-06
Phosphorylation of small GTPases has been also observed to affect binding affinity for the GDP/GTP cycle regulators notably GDP dissociation inhibitor (GDI) [10]. Indeed the EGF or cAMP-dependent phosphorylation of Cdc42 is associated with enhanced Cdc42–GDI interaction [8], [11]. RhoA inhibition by
-
br Cholesterol handling in the arterial
2020-01-06
Cholesterol handling in the arterial wall cells Foam cell formation in the arterial intima is a major hallmark of early-stage atherosclerotic lesions, which is attributed to uncontrolled uptake of modified lipoproteins, excess cholesterol esterification and impaired cholesterol release [44]. As a
-
br Experimental protocols br Acknowledgements
2020-01-06
Experimental protocols Acknowledgements This research was funded by the Croatian Ministry of Science, Education and Sports (Programmes 098-0982915-2948 and 098-0982522-2525), the Austrian Science Fund FWF (DK-MCD W1226, SFB LIPOTOX F30, P22832), and the Croatian-Austrian Intergovernmental S&T
-
Selective inhibitor of phosphodiesterase type PDE I
2020-01-06
Selective inhibitor of phosphodiesterase type 5 (PDE5I) is commonly used for ED treatment. PDE5I exhibits its vasodilatory effect by inhibiting the degradation of cyclic guanosine monophosphate (cGMP), which relaxes the smooth muscle and allows increased blood flow for penile tumescence. Udenafil (D
-
br Materials and methods F philippinensis plants
2020-01-06
Materials and methods F. philippinensis plants were collected in August 2014 at a farm in Ninning, Guangxi province, China and identified by Dr. Yimmin Zhao. Voucher specimens (No. 530) were deposited in the Herbarium of Gaunxi Botanical Garden, China. Human recombinant neutrophil elastase (EC 3.
-
Meropenem trihydrate mg The commonly used eDNA stains we
2020-01-06
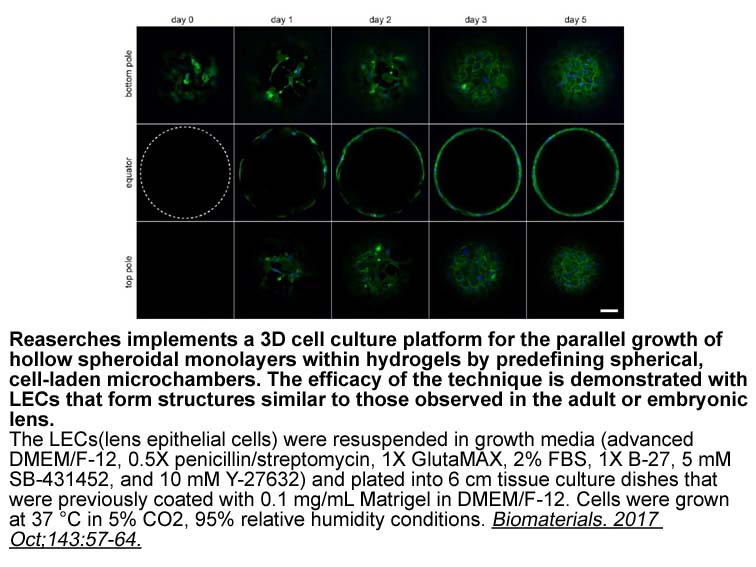
The commonly used eDNA stains we evaluated were the intercalating dye propidium iodide, the highly sensitive stain PicoGreen commonly used in DNA quantification, the cyanine monomer SYTOX® Green, and the dye 7-hydroxy-9H-(1,3-dichloro-9,9-dimethylacridin-2-one) (DDAO) not only sold as a standard for
-
CYP A is involved in the metabolism of approximately of
2020-01-03
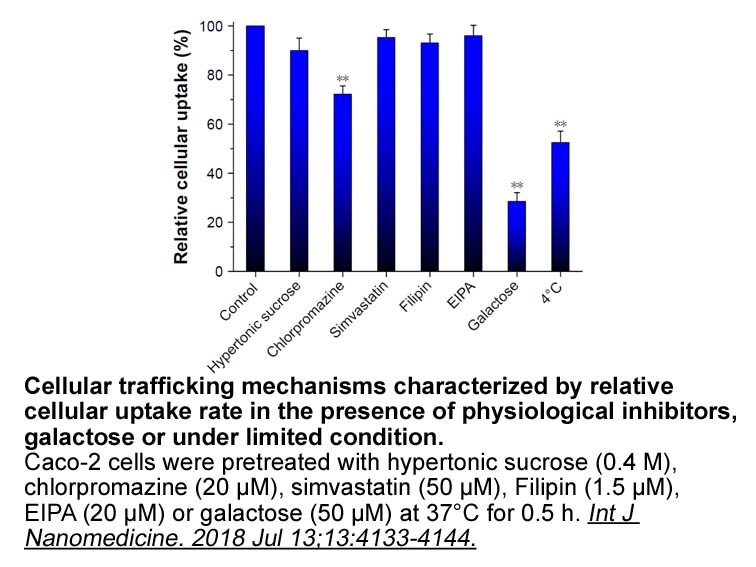
CYP3A is involved in the metabolism of approximately 50% of all drugs that are currently prescribed to patients [1], [15], [16]. Marked interindividual variabilitity of the catalytic function for metabolizing CYP3A substrates has been demonstrated with variability exceeding 20-fold [1]. Levonorgestr
-
br Author Contributions br Acknowledgments This work was
2020-01-03
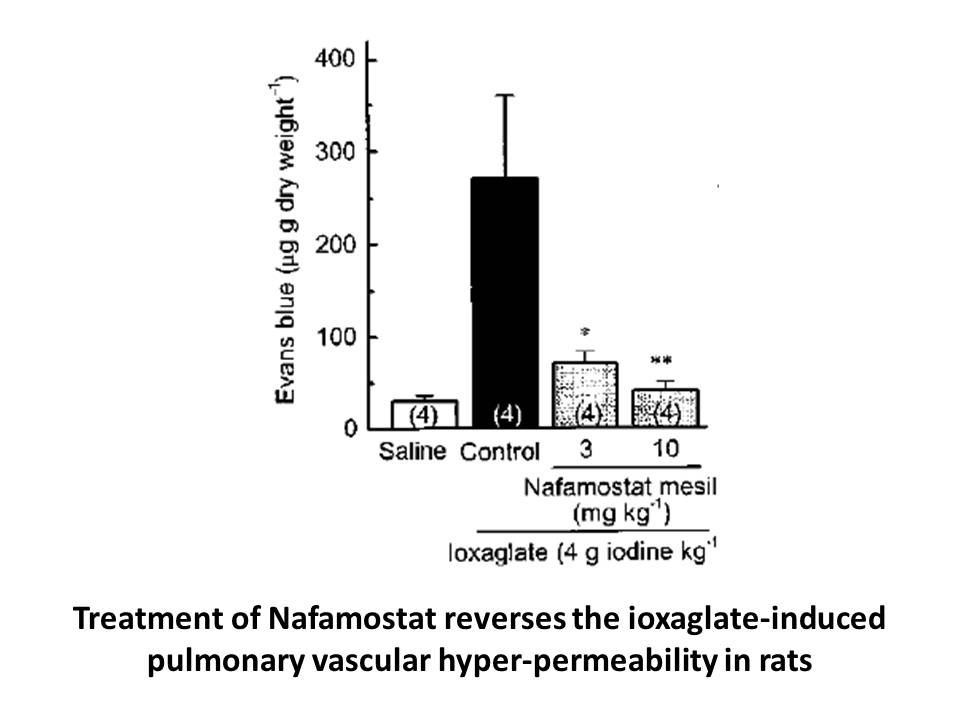
Author Contributions Acknowledgments This work was supported, in part, by a grant from the National Cancer Institute of the National Institutes of Health under Grant U01CA202241 and National Institutes of Health under Grant R01NS083856. Introduction The Eph (erythropoietin-producing human
-
Western blotting Western blotting was performed following th
2020-01-03
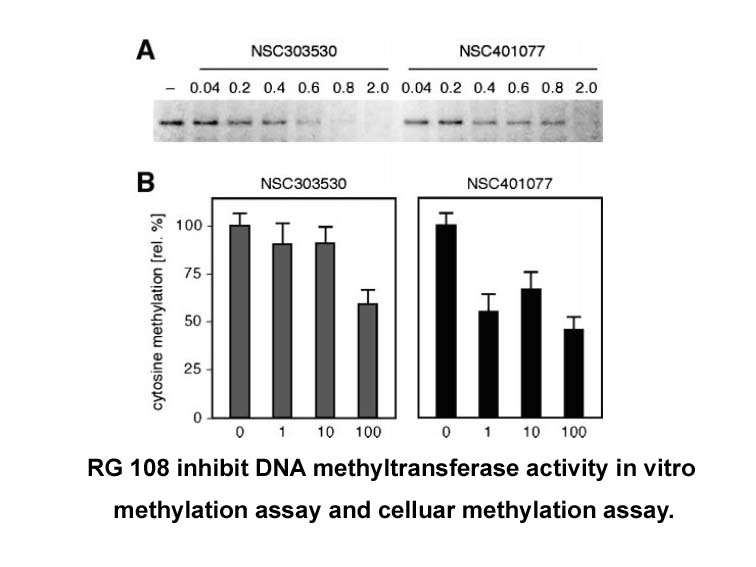
Western blotting: Western blotting was performed following the standard protocol as previously described [16]. All the images were collected using the Odyssey Fc imaging system (Li-COR Biosciences). In vitro kinase assays were performed on ∼1 μg of purified CDK5/p35 complex in 30 μL of kinase buffe
-
Placental insufficiency is regarded as the main etiology
2020-01-03

Placental insufficiency is regarded as the main etiology for FGR. Placental development is highly unique in eutherians and is regulated by numerous factors. For example, the novel retrotransposon-derived gene retrotransposon-like 1 (RTL1) was recently shown to play a key role in placental developmen
15852 records 893/1057 page Previous Next First page 上5页 891892893894895 下5页 Last page